Research in Biomedical
Problem Statement
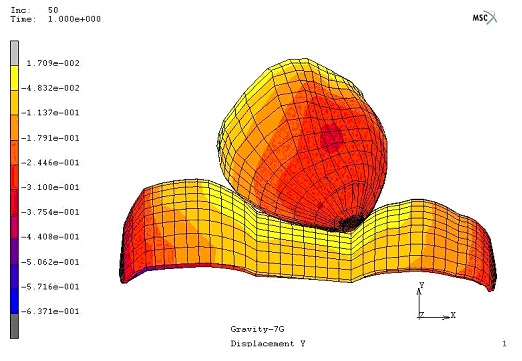
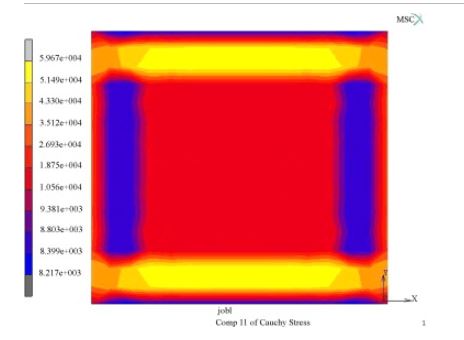
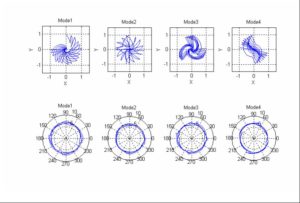
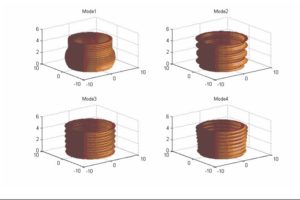
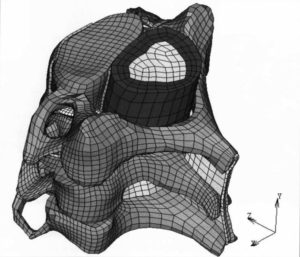
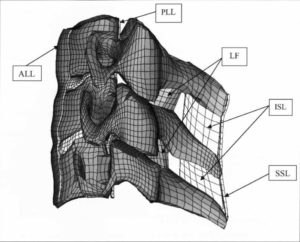
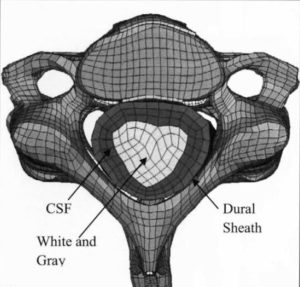
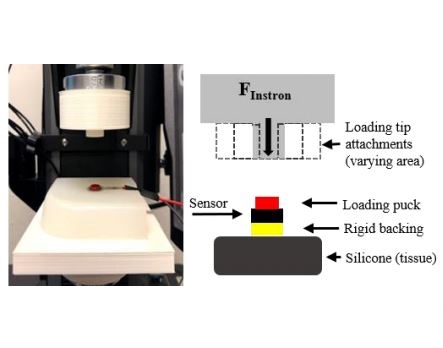
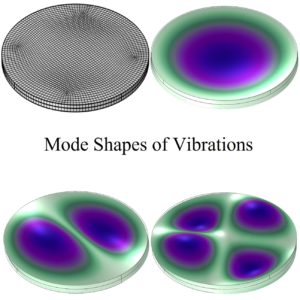
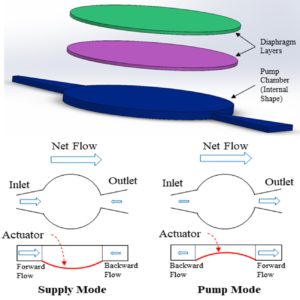
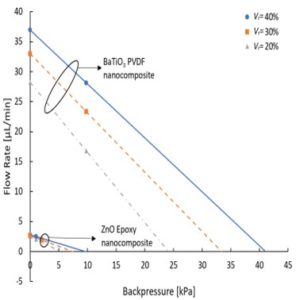
ARL-MLS is now navigating towards bridging the gap between biology and technology with the study of the various aspects of human body and medical care devices. The concepts are concerned with the impact of physical forces on biological structures and materials in the fields of fluid mechanics, solid mechanics, dynamics, and manufacturing, as well as its application to human health. Manufacturing encompasses the research and development of innovative processes for the fabrication of biomedical micro/macro devices, metal and polymer implants and sensors, tissue constructs, scaffolds as well as artificial organs. At ARL-MLS we strive to develop solutions for:
- Highly standardized and regulated methodologies.
- Human health automation.
- Space exploration and sustenance.
- Ergonomic and consumer friendly wearable devices.
- Energy scarcity for long term medical care products.
- Cost effective health care systems and methods for easy assimilation.
- Risk management and safety inspired device propagation.
- Novel Biocompatible material selection and adoption for higher functionality.
Solutions
To overcome the many global challenges in the field of Biomedical Research, ARL – MLS has been actively developing cutting edge solutions such as:
- High Precision interactive computational methods for real-time investigations in biomechanics.
- Regulatory Approach towards biomedical product innovation.
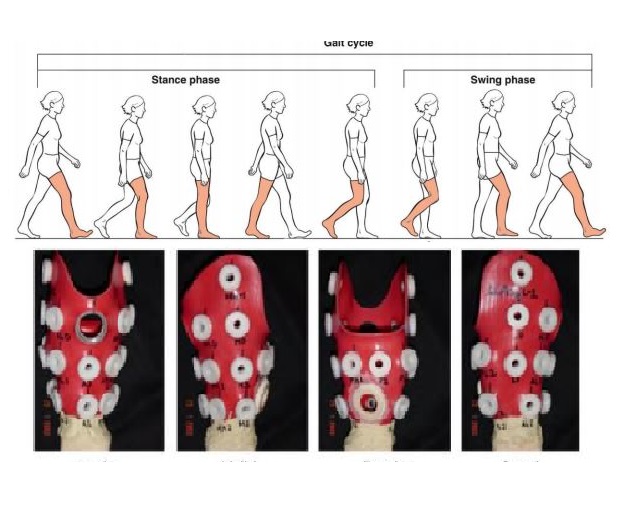
- Surrogate and smart models for live health monitoring and administration.
- Implantable and wearable solutions for sustainable health care infusing human factor studies.
- Energy solutions for wearable and implantable devices for long term application.
- Advanced Operation Research and Systems design methods towards biomedical product management.
- Interdisciplinary approach towards bioengineering solutions for improved user experience and product performance.
- Development according to industry best practices using ISO/IEC guidelines (ISO 13485, ISO 14971, IEC 60601, etc.).